Meet Our Microbiology Teams

The DSHS Microbiology Laboratory is one of the pillars of public health services in Texas. Each year it accepts and processes about 250,000 clinical and reference specimens from across Texas and its neighboring states. The work and dedication of each team in the Microbiology Unit contributes to protecting Texas communities and keeping Texans strong and healthy.
Each day, our testing services contribute to epidemiological investigations, disease diagnoses, disease monitoring and surveillance, preventative care planning, and public health policy development. Testing at the Lab identifies such conditions as:
- Respiratory, gastrointestinal, and sexually transmitted infections
- Foodborne and waterborne bacterial and viral infections
- Arboviral (mosquito-borne viruses) infections
- Mycobacterial and tuberculosis infections
- Rabies (animals only)
- Zoonotic infections
- Parasitic infections
- Fungal infections
Two major functions of the Laboratory are the diagnostic and reference testing of submitted specimens.
Diagnostic testing identifies whether a person is currently infected with or was previously exposed to a specific pathogen. This is done by analyzing a sample of blood, stool, serum, sputum, urine or other body fluid or tissue taken from the person. The majority of tests carried out at commercial and hospital laboratories are diagnostic. Diagnostic testing informs clinical care and is typically carried out in response to symptoms a person reports to their healthcare provider or signs the healthcare provider observes in their patient.
Reference testing is used to confirm diagnostic results or provide additional quantitative or qualitative data for complex cases. Most specimens received by the microbiology unit are submitted for reference testing. Reference testing supports outbreak investigations and allows the DSHS to monitor statewide and national infectious disease trends. This type of testing is often performed proactively, such as during screenings, in order to obtain data to identify trends in the spread of infectious diseases.
Pre-Analytical Teams
Receiving correctly labeled and appropriate specimens is an important factor in testing success at the Laboratory. Before a specimen can be processed, it must be logged and examined for its suitability to be tested. Members of the Check-In teams take on these important tasks every day.
Microbiology Check-In

Specimens are delivered, then logged and taken to the different Check-In areas to determine specimen acceptability. Photos © DSHS, 2024.
Our dedicated Microbiology Check-In Group is at the heart of the Laboratory’s operation. This group is first in line in ensuring the quality and safety of the Laboratory’s testing processes. Their work sets the foundation for accurate and reliable test results. Three teams make up the Microbiology Check-in Group: Bacteriology, Serology, and Texas Health Steps.
The Bacteriology Check-in Team manages the intake of a wide range of microbial specimens, from sputum specimens for TB testing, stool specimens for foodborne illness testing, to fungal specimens, and isolates submitted for infectious disease reporting purposes. They also receive mosquito surveillance samples that are tested for arboviruses.
The Texas Health Steps (THSteps) Team manages the intake of specimens submitted as part of the THSteps program, which provides children and adolescents with free medical checkups starting at birth until the age of 20 years. The THSteps program offers gonorrhea and chlamydia testing.
The Serology Check-in Team manages all serum and plasma specimen intake, and all specimens submitted for STI, HIV, and hepatitis testing. THSteps specimens for gonorrhea and chlamydia testing are processed by the Serology Check-in team
Opening mosquito shipping boxes to inspect the specimens in the Bacteriology Check-in area. Photo by DSHS, 2024. Cross checking patient identifiers on specimen labels and submission forms in a biosafety cabinet in the Bacteriology Check-In area. Photo by DSHS, 2024.
The Microbiology Check-in team has responsibilities for:
Package Inspection: The team carefully inspects and opens delivered packages. Some microbial specimens that are submitted to the Laboratory can pose serious health risks. The Check-in Team handles them carefully. For example, tuberculosis (TB) sputum specimens are opened inside a biosafety cabinet. Using biosafety cabinets ensures both the specimens and the Check-in Team members are protected from possible contamination.
Quality Checks: To preserve specimen integrity, the team verifies that the correct specimen types are received for the tests requested and that they meet all standards required for testing.
Documentation Verification: The team reviews the specimen submission forms and all accompanying paperwork to confirm their accuracy and ensure every detail is correctly recorded.
Verifying Accurate and Usable Labeling: The team ensures each specimen label matches its submission form. Once a match is confirmed, Check-in Team members use basic demographic information to create an internal Sample ID and barcode for each specimen in the Laboratory Information Management System (LIMS). This crucial step supports specimen tracking and management throughout the Laboratory. The Check-in Team then sends specimen submission forms to the Data Entry (DE) Team. The DE Team finishes uploading the demographic information and any additional paperwork into the LIMS. Documentation discrepancies are addressed by the Microbiology Specimen Logistics Team to ensure accurate records and testable specimens.
Safety Protocols: -The Check-In Team follows strict safety protocols to manage and isolate any hazardous materials, safeguarding our lab environment.
Our Microbiology Check-in Team’s work is essential in maintaining the Laboratory’s high standards. Their dedication is a key part of delivering reliable and accurate test results to our submitters.
Microbiology Specimen Logistics

A member of the Microbiology Specimen Logistics Team reviewing forms.
Photo by DSHS, 2024.
All specimens must have the correct information and documentation with them before they can be tested. The Microbiology Specimen Logistics Team plays a crucial role in managing the acceptability of specimens that arrive at the Laboratory without correct information. They often work alongside the Check-in Teams to address submission issues.
Have you ever been called by the Laboratory with a request for more specimen information or to update or correct specimen submission paperwork? The Microbiology Specimen Logistics Team contacts submitters to collect missing information and troubleshoot documentation issues that could delay or prevent testing. The team also notifies submitters when specimens are not acceptable for testing and if a specimen should be recollected and resubmitted. They also make courtesy calls to rabies specimens submitters to notify them of negative test results.
Lastly, the Microbiology Specimen Logistics Team reviews internal testing statistics for quality assurance and process improvement purposes. Through their customer service and commitment to high quality service, in 2024, over 4,400 specimens were saved from rejection. In doing so, countless hours of additional work for patients, providers, and Laboratory staff are saved.
Laboratory Supply, Shipping, and Container Preparation

A member of the Container Preparation Team gathering supplies to be sent out to a submitter. Photo by DSHS, 2024
Container Preparation Team

Boxes of supplies ready to be shipped to submitters. Photo by DSHS, 2024
The Container Preparation Team plays a vital supporting role in microbiology testing for several public health programs. It provides specimen collection kits and shipping supplies to healthcare providers who participate in programs including antimicrobial resistance testing, Candida colonization, Texas Infertility Prevention Program, HIV and STD Prevention Program, Influenza Surveillance, and the Tuberculosis Prevention Program

The container preparation workspace with program testing and shipping supplies. Photo by DSHS, 2024
The team also reviews orders via email or fax, checks order history to avoid duplicates, ensures collection kits are not expired, and contacts submitting facilities for clarification on order issues.
External customer service is a key aspect of their role, as they are often the first point of contact for troubleshooting issues related to specimen collection kit requests, billing holds, and shipping information. They also guide submitters to the correct program areas for additional assistance. The work of the Container Preparation Team is vital in maintaining the supply of specimen collection kits to eligible submitters. Accurate and timely order processing is critical to receiving testable specimens since delays can hinder submitters' ability to perform essential screening services for patients.
Laboratory Supply Team
The Laboratory Supply Team provides everything from general consumables such as gloves to reagents for the Laboratory. It ensures that supplies are there for the testing teams when they need them. The Laboratory’s testing schedules and operations depend on the Supply Team’s work. Their efficiency means that specimen processing is reliable, and testing runs on time.
Data Entry & Demographics and Laboratory Reporting Groups
Each year, the Data Entry & Demographics and Reporting Groups enter the demographic data for approximately 1.5 million specimens into the LIMS for all of the Laboratory’s Units. The groups support the Laboratory before testing begins and after it is completed, and they wear many hats!
The Data Entry Group creates and maintains submitter accounts for new and existing submitters. They also provide submitters with specimen submission forms. Once the Serology and Bacteriology Specimen Check-in Teams are finished checking newly arrived specimens, the Micro Data Entry & Demographics Group enters demographic information from the submission forms while the various Laboratory’s test the specimens.
If documentation errors prevent a specimen from being tested, the Micro Data Entry & Demographics Group will coordinate with the Microbiology Specimen Logistics Team to gather the missing information from the submitter and make the necessary corrections to the specimen information in the LIMS.
After testing is complete, the Laboratory Reporting Group checks and finalizes the demographic information that was reviewed by the Check-in teams. The team also uploads any additional or required paperwork for specimens to the LIMS. After the test results are posted to the LIMS, a final test results report is released to the specimen submitter. Submitters can choose to receive their results electronically, by fax, or via mail.
Laboratory Testing Teams
Arbovirus/Entomology

The Arbovirus/Entomology Team Photo by DSHS, 2024
 A mosquito collection carton with mosquitoes. Photo by DSHS, 2024
A mosquito collection carton with mosquitoes. Photo by DSHS, 2024
The Arbovirus/Entomology Team carries out the vector monitoring system for the Texas Arbovirus Surveillance Program. The goal of the program is to assist local jurisdictions in Texas with their Integrated Mosquito Management activities by providing mosquito species identification, arbovirus testing, and insecticide resistance (IR) testing services.
The team carries out routine mosquito testing from May through November each year, with some flexibility available for outbreak situations or cases of arboviral diseases. Mosquito identification and IR testing services are provided year-round.
The Arbovirus/Entomology Laboratory typically receives between 10,000-15,000 mosquito trap collections each year. The team tests the 200,000 to 500,000 mosquitoes taken from these traps for arboviruses including West Nile virus, St. Louis encephalitis virus, and eastern equine encephalitis virus.
The Arbovirus/Entomology Laboratory typically receives between 10,000-15,000 mosquito trap collections each year. The team tests the 200,000 to 500,000 mosquitoes taken from these traps for arboviruses including West Nile virus, St. Louis encephalitis virus, and eastern equine encephalitis virus
A person in a labcoat looking through a microscope at insects.
Clinical Bacteriology

The Clinical Bacteriology Group Photo by DSHS, 2024
The Clinical Bacteriology Group tests clinical specimens and isolates that are submitted to the Lab to comply with Texas Notifiable Conditions requirements (Texas Administrative Code Chapter 97). The group also confirms or completes the findings of other laboratories

Clinical Bacteriology Group processing samples in biosafety cabinets and on the bench. Photo by DSHS, 2024
that do not have the resources to carry out specialized procedures on reportable isolates. Assisting epidemiologists and other Lab testing areas with epidemiological and surveillance studies is another important role the group fills.
.png?cb=1743714766)
Loading software for MALDI testing. Photo by DSHS, 2024
The group identifies bacteria from clinical specimens and pure isolates using several methods, including matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-ToF), conventional biochemicals, phenotypic methods, commercial test kits, gas-liquid chromatography, and serogrouping or serotyping. The isolates obtained by the Clinical Bacteriology Group from clinical specimens are commonly sent to the Genetic Sequencing Branch for sequencing
Clinical Virology

The Clinical Virology Team Photo by DSHS, 2024
The Clinical Virology Team performs molecular testing using real-time RT-PCR to identify viruses of public health importance from clinical specimens. The team works closely with state epidemiologists to provide testing services for diseases such as measles, mumps, and arboviral infections in people caused by dengue, chikungunya, and Zika viruses. These test results assist with case and outbreak investigations. The Clinical Virology Team also performs testing to assist with influenza and COVID19 surveillance efforts within the state and carries out real-time PCR detection of Rickettsia bacteria (yes, bacteria!) that cause Rocky Mountain spotted fever and typhus.

Filling the centrifuge to prepare for PCR analysis. Photo by DSHS, 2024
Consumer Microbiology

The Consumer Microbiology Group Photo by DSHS, 2024
The Consumer Microbiology Group tests drinking (or potable) water, food, shellfish, and milk and dairy products for the presence of infectious organisms such as Shiga toxin-producing Escherichia coli, Salmonella, Listeria, and Vibrio bacteria.
The group partners with the Texas Commission on Environmental Quality in testing drinking water samples from municipalities across Texas for the presence of coliform bacteria and E. coli. It also accepts well water samples from the public for coliform and E. coli testing.

Left to right: Testing potable water for coliforms, and weighing prepackaged jerky to add to a paddle blender (AKA stomacher). Photo by DSHS, 2024
The group routinely accepts food samples submitted by registered sanitarians for regulatory testing and in cases of foodborne disease outbreaks. The DSHS Seafood and Aquatic Life Group routinely collects and submits shellfish samples to the Laboratory for testing to monitor the quality of the Texas bay waters where oysters are harvested. DSHS Milk and Dairy Unit sanitarians submit milk and dairy products to the Lab for testing to meet national regulatory process testing requirements.
Sanatarians also submit milk and dairy products following complaints or outbreaks. In cases of foodborne outbreaks, registered sanitarians from the DSHS Consumer Protection Division or local health departments submit unprocessed and “Ready to Eat” foods directly to the Laboratory for testing.
In 2023, the Consumer Microbiology Group tested approximately 2,000 food samples, 2,100 shellfish samples, 800 milk and dairy samples, and 850 water samples.

Streaking a plate for bacterial growth. Photo by DSHS, 2024
Serological Analysis Group

The Serological Analysis Group Photo by DSHS, 2024
The Serological Analysis Group consists of two teams: the Serology Team and STD Team.
The Serology Team carries out serologic testing on clinical specimens to detect infections caused by viruses, bacteria, and parasites. Serologic tests work by detecting specific antibodies the body’s immune system makes when a person is infected with a pathogen. The Serology Team works with state epidemiologists to provide diagnostic testing that helps with case and outbreak investigations. Examples of infections the team helps detect in patients include measles and mumps. The team also helps diagnose less common infections including hantavirus pulmonary syndrome, brucellosis, and several rickettsial and arboviral infections. The Serology Team also provides parasitic infections screening services for the medical screening program run by the U.S. Committee for Refugees and Immigrants (USCRI).
The STD Team provides screening and diagnostic testing services for sexually transmitted infections (STIs). These include chlamydia, gonorrhea, hepatitis C virus (HCV), human immunodeficiency virus (HIV), and syphilis. The team also tests specimens from patients who are being treated for STIs. These tests help to track the effects of HCV, HIV, and syphilis treatments on the patients’ infections. The STD Team also provides STI screening services to submitters who are approved to take part in certain DSHS-run prevention programs. These programs, which offer STI testing include the Texas Infertility Prevention Project (TIPP), Texas Health Steps, and the HIV/STD Prevention Program.
Left to right: Manually agitating a test tube for syphilis rapid plasma regain (RPR) testing and loading samples onto the Panther. Photos by DSHS, 2024
Genetic Sequencing Branch

The Genetic Sequencing Branch. Photo by DSHS, 2024
The Genetic Sequencing Branch is the newest testing lab at the DSHS Austin Laboratory. It has two distinct teams: Advanced Molecular Detection and Microbial Genomics.

Two NovaSeq 6000s in the Sequencing Room Photo by DSHS, 2024
The Advanced Molecular Detection (AMD) Tam prepares, analyzes, and compares the whole genomes of microbes obtained from food samples, environmental samples, clinical specimens, and pure isolates. The team analyzes the whole genome data of pathogens for helpful information such as serotypes, subtypes, mutations of interest, or toxin genes. The team then uploads these data to the CDC’s national enteric database to share with other researchers across the country and rest of the world. Once uploaded, the genomes are available for analysis to investigate the role of the pathogens in foodborne disease cases or clusters. The AMD Team also conducts phylogenetic analysis on sequence data of foodborne organisms to identify the relatedness of specimens involved in outbreaks. Phylogenetic data helps epidemiologists in outbreak investigations.
The Microbial Genomics Team carries out targeted and whole genome sequencing (WGS) of pathogens from clinical and environmental samples, including SARS-CoV-2, Human monkeypox virus, Cyclospora, Influenza virus, and Mycobacterium tuberculosis. In 2022, the team began sequencing surveillance of wastewater for SARS-CoV-2 through an FDA pilot project. As part of the wastewater project, the team collaborates with DSHS epidemiologists and an academic partner. The wastewater surveillance program continues to grow as new water sampling sites and organisms of interest are identified. The team is also working on a metagenomics sequencing project with the CDC and APHL to identify pathogens in different types of clinical specimens such as whole blood, serum, lesion swabs, and stool.
The team will soon expand beyond sequencing microbes: They are performing preliminary testing for the targeted sequencing of genes that cause newborn screening disorders to identify sequence variants that cause disease.

Preparing gene libraries for sequencing. Photo by DSHS, 2024
Molecular Microbiology

The Molecular Microbiology Team. Photo by DSHS, 2024
The Molecular Microbiology Team (MMT) supports testing for infectious disease and foodborne pathogen samples that are received by the Clinical Bacteriology Group, the Zoonotic Diseases Branch, and the Environmental Microbiology Branch. The team carries out a variety of testing methods as part of their analyses, such as real time PCR, nested PCR, conventional PCR, gel electrophoresis, Sanger sequencing, and manual and automated nucleic acid extractions.

Working in the PCR room Photo by DSHS, 2024
The MMT identifies a variety of pathogenic bacteria, viruses, and parasites, including Norovirus, Plasmodium, Cyclospora, Cryptosporidium, Shiga Toxin Producing Escherichia coli (STEC), and Carbapenem Resistant Organisms (CROs). In doing so, the team collaborates with the CDC in a variety of projects such as CaliciNet (Norovirus genotyping), CryptoNet (Cryptosporidium subtyping), Cyclospora AMD (Cyclospora genotyping), and ARLN (Antimicrobial Resistance Laboratory Network).
The MMT also performs nucleic acid extractions to support the sequencing activity of the Genetic Sequencing Branch. Their analyses greatly assist state and local epidemiologists during outbreak investigations and responses. The team also collaborates with the FDA under multiple federal grant supported projects.

Loading plates for extraction before they are sent for whole genome sequencing and PCR. Photo by DSHS, 2024
Mycology
Left to right: Reviewing organism growth under the microscope and preparing culture tubes. Photo by DSHS, 2024
The DSHS Mycology Team receives referred isolates of yeast, mold, and aerobic actinomycetes for identification from clinical and reference laboratories across Texas. The team identifies mycological cultures based on macroscopic and microscopic morphology, and other characteristics of the isolates and their reactions to a panel of biochemical tests. Identification of most molds relies on microscopic morphology. Yeast identification relies on biochemical reactions, MALDI-ToF testing, and microscopic characteristics. The team’s turnaround time for most referred mycological cultures ranges from two to four weeks, depending upon the number of tests required for identification.
Medical Parasitology

Observing slides under a microscope together. Photo by DSHS, 2024
The Medical Parasitology Team may be the smallest in the Laboratory, but they are kept busy! The team processes and examines whole blood, urine, stool, and individual worms, to detect and identify parasitic infections in humans. As the public health reference laboratory for the state of Texas, the team coordinates with state and local epidemiologists and the CDC in the surveillance of medically important parasitic diseases. The Parasitology Team also routinely examines specimens collected by the medical screening program of the Texas Refugee Health Program, which is now administered by the U.S. Committee for Refugees and Immigrants.

Preparing washes for slide staining. Phot by DSHS,2024
Rabies

Left: Examining a brain specimen for rabies testing. Right: A (deceased) bat specimen in a sealable, see-through bag, which is the preferred method of submitting small, bat-sized mammals. Photo by DSHS, 2024, 2023
The Rabies Laboratory at DSHS in Austin is the primary rabies diagnostic laboratory in the state of Texas. It also serves as a rabies reference laboratory for other laboratories in Texas, as well as for nearby states.
The Rabies Team receives between 7,000 to 9,000 animal specimens each year for testing (approximately 27 to 35 specimens every day), so the lab is very busy! The team receives mostly domestic animals such as cats and dogs for testing, but the highest positivity rate in specimens is found in wild animal specimens, particularly skunks and bats. The team's testing procedures are designed to provide rapid reporting of results despite the volume of specimens tested daily. Results are usually available 24–48 hours after receipt of a specimen, except on weekends.
Mycobacteriology (Tuberculosis)

The TB Diagnostics Team. Photo by DSHS, 2024
The DSHS Austin Mycobacteriology Laboratory is one of the largest in the country. It consists of three teams: TB Diagnostic Team, TB Analysis Team, and TB Reference Team. The tuberculosis (TB) testing services offered by the Mycobacteriology Laboratory are twofold:
- Diagnostic testing
- Reference testing

Loading samples onto the MGIT. Photo by DSHS, 2024
The TB Diagnostic Team identifies Mycobacterium species and other aerobic actinomycetes from submitted clinical specimens and referred isolates. The TB Analysis Team carries out susceptibility testing for several antimicrobials on Mycobacterium tuberculosis complex (MTBC) and Mycobacterium kansasii. Once a specimen is confirmed as containing M. tuberculosis, it is sent to the TB Reference Team, where the identification is confirmed as M. tuberculosis or another mycobacterium.
The TB Reference Team also tests the drug susceptibility of confirmed MTBC and M. kansasii isolates. The Analysis and Reference teams may collaborate with medical providers who are part of the Tuberculosis Elimination Program in treating patients infected with multiple drug resistant M. tuberculosis. The Tuberculosis Elimination Program is one of the highest volume TB specimen submitters to the Laboratory.
%20and%20observing%20slides.png?cb=1743714766)
Examining tubes for fluorescence (left) and observing slides under a microscope (right). Photo by DSHS, 2024
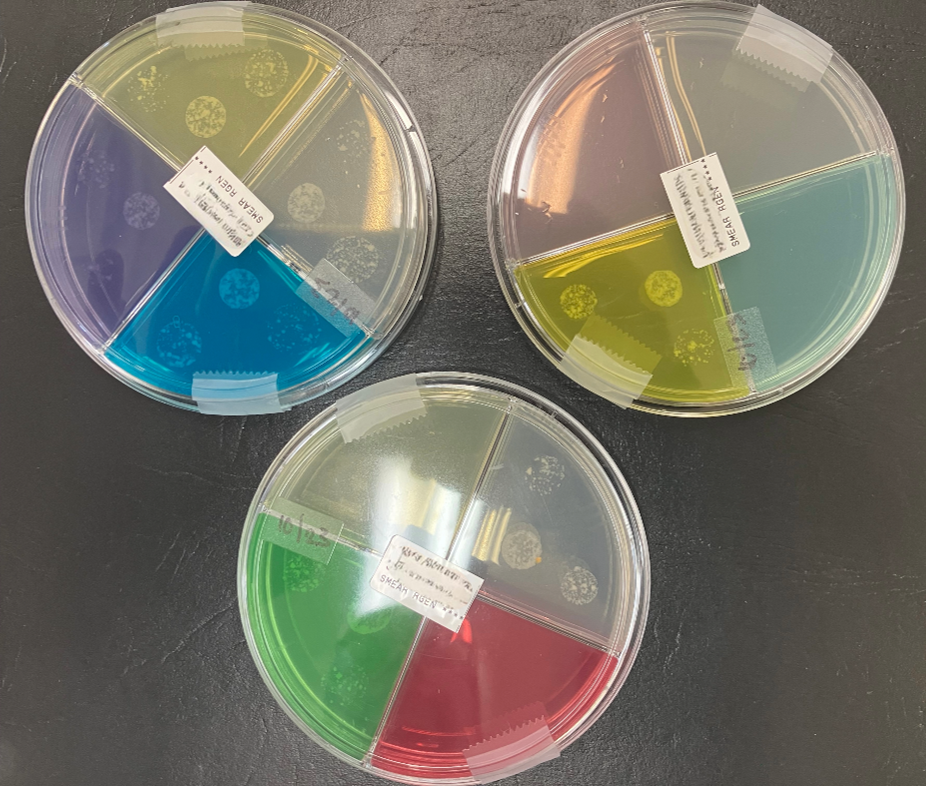
Quadrant plates with selective M. tuberculosis growth. Photo by DSHS, 2024
Essential Support Teams
The Microbiology Unit has several support teams that ensure specimen processing and analyses go as smoothly as possible
Education Specialists

Preparing for presentations at the Laboratory Connect Conference. Photo by DSHS, 2024
The Microbiology Unit’s education specialists are a key link between the Unit and our submitters and other external customers. A major role of the educators is to help reduce the number of unsatisfactory specimens the Lab receives by educating or training submitters on the Lab’s specimen submission requirements. The educators also help demystify or troubleshoot other submission and testing processes by answering questions from our submitters and other external customers. Graphics-based specimen submission job aids created by the educators, available here, help submitters avoid common causes of specimen rejection.
The Microbiology educators attend local and national conferences to present and share educational and training materials with attendees and current or potential submitters to the Lab. They also routinely update submitters on changes to testing and submission requirements via the GovDelivery email distribution system and update the Microbiology Unit’s webpages as needed.
The educators spotlight the role of the Laboratory in Texas’ public health system by conducting tours of the Laboratory for various visitors, including students, first responders, lawmakers, and other stakeholders. These Lab tours help showcase job opportunities at the Lab as well as ways for customers to collaborate with the Lab’s testing teams.

Presenting at a conference for AR Lab Network activities. Photo by DSHS, 2024.
Informatics Branch
The DSHS Public Health Laboratory Informatics Branch consists of the LIMS and Informatics Group and the Bioinformatics Team. The Branch fulfils several emerging roles in electronic medical data transfer and management.
The LIMS and Informatics Group works with the IT-Lab Applications Development Team to administer the Microbiology Unit’s Laboratory’s Information Management System (LIMS). The group also develops solutions for data extraction and transfer from the LIMS.
Informatics manages the exchange of information with multiple recipients of Lab data, including CDC and the National Electronic Disease Surveillance System (NEDSS). In doing so, the group ensures epidemiological data for samples tested by the Laboratory and its partners are shared reliably and smoothly. Through this work, the group helps to inform the public health staff and key decision makers who monitor disease levels in their communities. The data managed by the Informatics Group includes Laboratory information on over 100 different infectious diseases that have the potential to trigger large scale outbreaks if not identified and controlled early on.
The Bioinformatics Team manages and processes the data produced by the Genetic Sequencing Branch (GSB), which obtains specimens for sequencing from the DSHS Lab, university collaborators, and occasionally from CDC. Once these specimens are sequenced by GSB, the Bioinformatics Team manages and processes the data.
These data are vital in disease outbreaks and the Bioinformatics Team makes them available to submitters, epidemiologists, and CDC or FDA to help with investigations.
The team also assists in managing the data for uploading to NCBI’s Sequence Read Archive, Genbank, and GISAID, among other databases. Once in these databases, the data are public and available to researchers across the country and world. The scope of sequencing work at the Laboratory is growing each year, so the impact of the Informatics Branch will also grow over time.
Media Preparation

Various stages of media development, from preparation to storage. Photo by DSHS Lab, 2024.

The Media Prep Team after dispensing broth into tubes and jars. Photo by DSHS, 2024
The Media Preparation Team is the backbone of the Lab in that it produces most of the Laboratory’s growth and testing media. In addition to producing commonly used media like MacConkey Agar and Tryptic Soy Broth, the Media Prep Team helps testing labs develop unique media and other products that cannot be purchased commercially.
Each year, the team produces approximately 500,000 units of media from over 430 active recipes that include buffers, indicators, enrichment broths, selective media, agar slants, agar plates, and other media, which helps the lab save costs. Media Prep also quickly prepares media for testing areas in response to outbreaks. For example, they produced media for several Listeriosis outbreaks here in Texas and helped produce and prepare commercial viral transport media for testing during the COVID-19 pandemic.
.png?cb=1743714766)
Left to right: Measuring agar, and dispensing agar onto plates. Photo by DSHS, 2024